
Tackling society’s current and future challenges with Supercomputing Applications:
The European Centres of Excellence in HPC
From computing the reduction of noise and fuel for passenger airplanes to assessing the effects of climate change – applications in high-performance computing (HPC) help tackle some of the world’s biggest societal, scientific, and economic challenges. The Centres of Excellence (CoEs) for High Performance Computing (HPC) applications presented on this website develop such applications and have the potential to help society deal with current and developing concerns:
- A number of the CoEs were in a position to contribute to the fight against the pandemic (featured in our newsletters and in the special website contribution).
- HiDALGO’s migration modelling application targeted the capacity to support organisations like Save the Children in analysing and reacting to forced human migration.
- Many of the CoEs have the potential to contribute to the European objective of reducing global dependencies through their HPC-enabled tools supporting the development of new technologies and solutions, for example in the areas of renewable energy and new materials.
The development of the next generation of supercomputing systems – Exascale systems, with computing speeds a billion times higher than current desktop computers – targets the future needs of such applications, and is the goal of the European Union and the EuroHPC Joint Undertaking. In 2020-2021, Petascale systems and the first pre-Exascale systems of the EuroHPC Joint Undertaking started to come online.
Enabling these applications to effectively use ever larger (and increasingly complex) computing systems is a major milestone and a central goal of the European HPC CoEs. After an investment of 42 million Euro in 2015, the European Commission confirmed its support with an additional budget of 74 million Euro in December 2018 resulting in 10 operational European HPC CoEs for computing applications. Between October 2020 and January 2021, five more CoEs officially started their activities. Achieving excellence in HPC Applications takes a pivotal place – it is the applications that deliver solutions to the aforementioned challenges – in the European HPC Ecosystem alongside the development of Exascale technologies and provision of access to extreme-scale infrastructure.
In January 2023, 10 further CoEs started their activities, and two more in January 2024, all with a clear focus on porting their codes to the growing set of high-performance computing systems of the EuroHPC JU.

Concerted action for the European HPC CoEs
In order to maximize the impact of the CoEs, Coordination and Support Actions (CSAs) have been funded order to support the CoEs in their mission to contribute to a globally competitive HPC ecosystem.
The more recent CSA is CASTIEL2, running from January 2023 until December 2025. Its mission is to support both CoEs and the National Competence Centres (NCCs) in reaching their goals and to establish and nurture close exchange and mutual benefit between these projects. CASTIEL2 has received funding from the European High-Performance Computing Joint Undertaking (JU) under grant agreement No 101102047. The JU receives support from the Digital Europe Programme and Germany, Italy, Spain, France, Belgium, Austria.
Its forerunner project FocusCoE ran from December 2018 to March 2022 and contributed to the success of the European high-performance computing “ecosystem”. The project supported the Centres of Excellence in high-performance computing to more effectively fulfil their role, which is to ensure that extreme scale applications result in tangible benefits for addressing scientific, industrial or societal challenges.
While FocusCoE ended, the CoEs continue their work and will continually benefit from the assets that FocusCoE has created. This website and related activities in the social media will stay online and continue to be a central hub for past, current and future European HPC CoEs. FocusCoE partner University of Stuttgart has pledged to offer pro-bono support for this task.
FocusCoE has initiated the foundation of the HPC CoE Council (HPC3), a body comprising all active HPC CoEs and aiming at strengthening the role of applications in the European HPC ecosystem, and has supported its operations. This included establishing contacts with a number of relevant players in the European HPC ecosystem. HPC3 will continue to operate after the end of FocusCoE, supported pro-bono by the FocusCoE coordinator scapos AG. It is hoped that the next generation of CoEs funded through the EuroHPC Joint Undertaking will also participate in HPC3.
FocusCoE has supported the CoEs in a multitude of ways, beyond HPC3: It has facilitated industrial outreach, organized transversal trainings, analyses and guides for the CoEs, compiled the individual offerings of the CoEs and presented them in a way useful for external users, and has created and propagated an overarching “CoE brand” in order to disseminate widely the fascinating results and achievements and more broadly the contributions of HPC to society, science and industry.
Beginning with the “CoE brand”, FocusCoE created the HPC CoE web site hpccoe.eu as central hub for all CoE-related news, results, and offerings.
- CoE use cases and industrial success stories are collected and presented
- News from the CoEs are regularly presented on the website, and 15 newsletter issues have been compiled and sent out
- The offerings of the CoEs have been compiled and presented in several categories, most notably software, consulting and training. The web site provides a variety of ways for potential users to search for CoE-supported software that could fit their needs
- In collaboration with PRACE and CASTIEL, a pan-European HPC training registry has been developed which suits the extended needs of the CoE training offers, and the hpccoe.eu web site is the unique place to access all CoE trainings
- A calendar of events of interest for the CoEs is maintained, with a particular focus on events with CoE participation.
- The many videos of tutorials and webinars of the CoEs have been organised in various playlists on FocusCoE’s YouTube channel
- FocusCoE has disseminated CoE related news over Twitter and LinkedIn
- FocusCoE compiled an overview of innovations emerging from the Centres of Excellence.
Beyond that, FocusCoE has created thematic overview articles to put the work of the CoEs into a broader context: One article covering COVID-19 related research and another one on climate change research. Addressing a broad audience beyond the technical and scientific sub-communities is an overarching goal for FocusCoE, and it was a driving force behind the compilation of the CoE brochure released towards the end of FocusCoE which targets the informed general reader.
FocusCoE was instrumental or has contributed to organizing public events and workshops:
- A pan-European HPC Training Stakeholder Workshop on 8th Oct 2019 in Brussels, organised in conjunction with the European Commission, attracting 32 participants
- A follow-up workshop at the EuroHPC Summit Week 2021, “HPC Education & Training – Perspectives from EU13 Member States”, focused on the HPC education and training landscape among the EU13 Member States and attracted about 100 online participants
- A workshop at the EuroHPC Summit Week 2021 “CoEs on the road to Exascale” with an introduction of FocusCoE, an overview over the CoEs and their applications, and a panel discussion, attracting ca. 100 online participants
- A workshop at the EuroHPC Summit Week 2022, “European HPC CoEs: perspectives for a healthy HPC application eco-system and Exascale”, featuring an introductory talk and two round table discussions with participation of all active CoEs
In addition, FocusCoE organized several joint participations or booths in general HPC-related events, for instance the Teratec Forum 2021 or ISC 2021.
In order to support the industrial outreach of the CoEs, FocusCoE has organized a series of joint visits to sectorial industrial events. Only the first one (BIOFIT2019, Dec. 2019) was a classical event, all the others were hit by the restrictions of the pandemic and eventually took place online. FocusCoE has developed with the CoE best practices to get most out of these online as well as in-person industrial events. In addition, FocusCoE has organized thematic webinars with industry as an alternative format (Opportunities and Challenges for Industrial Applications” in February 2021, and another one during the HIPEAC sessions: “Industrial Challenges and Innovation Opportunities of CoEs in Medical/Bio/Pharma sectors“ in May 2021). In addition, FocusCoE organised workshop on specific challenges of interaction with SMEs, and carried out a survey on industrial needs as well as an analysis of the CoE’s innovation and IP management, both resulting in valuable material for the CoEs.
A considerable number of training courses, workshops, materials and guidelines have been organized or created for the CoEs, to support them with cross-cutting aspects of their work. Beyond those already mentioned, these were:
- Training courses on pedagogy / training methodology
- Training courses on research commercialization
- Workshops on business & sustainability and co-design aspects
- Analysis of CoE training offers
- Template training materials for trainings on running HPC applications at scale
Frequently Asked Questions
High-performance computing (HPC) refers to systems with extremely high computational capabilities. They involve hundreds of thousands of processors working in parallel to analyse billions of pieces of data in real-time. Today’s most powerful systems can perform calculations thousands of times faster than a normal desktop computer. The European Union has recognised high performance computing as a key component of the digital single market strategy.
High-performance computer simulations provide important insights to predict the behaviour of processes and products in many areas of science and engineering. It provides important tools, e.g. to the automotive industry, aerospace, biotechnology, environmental science science including climate research, medical technology, intelligent manufacturing, engineering among many others. The fastest computers in the world today are already incredibly powerful. Nevertheless, many scientific and industrial challenges demand still more computing power, for instance bioscience, engineering or material design.
Over the last decades, the availability and usability of HPC systems has become one of the determining factors for the progress of science and a strategic resource for Europe’s future. The accuracy of the analysis, the quality of the results and the potential of national and international collaborations in science and industry are more and more depending on the available computing infrastructure. Mastering advanced computing technologies from hardware to software has become essential for innovation, growth and jobs.
Over the last decades, the availability and usability of HPC systems has become one of the determining factors for the progress of science and a strategic resource for Europe’s future. The accuracy of the analysis, the quality of the results and the potential of national and international collaborations in both science and industry are more and more depending on the available computing infrastructure. Mastering advanced computing technologies from hardware to software has become essential for innovation, growth and jobs.
For the European Commission, HPC needs an EU-level policy to optimise national and European investments, addressing the entire HPC ecosystem. Therefore, its HPC Strategy was adopted in 2012 and implemented in the biggest EU Research and Innovation programme Horizon 2020.
Based on contractual Public-Private Partnership on HPC (cPPP on HPC), a strong cooperation with the HPC stakeholders is set up and developed an ambitious Research & Inovation funding strategy, addressing the following aspects:
- HPC facilities via the Partnership for Advanced Computing in Europe (PRACE), established since October 2012
- HPC technologies via the Future and Emerging Technologies (FET) programme, represented by the the European Technology Platform for HPC (ETP4HPC)
- and HPC applications via the establishment of Centres of Excellence (CoEs); the CoEs are funded via the e-infrastructures programme which aims to address the needs of European researchers for digital services in terms of networking, computing and data management.
These three pillars are building the European HPC Ecosystem.
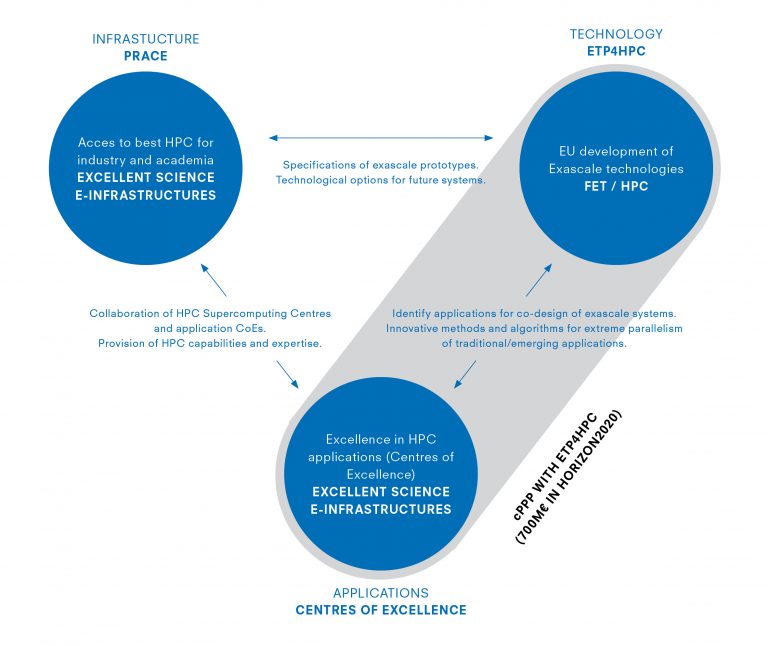
The European Centres of Excellence (CoEs) for high-performance computing are promoting the use of upcoming exascale and extreme performance computing capabilities, prepare the existing parallel codes for this next generation of supercomputers and provide e.g. consulting and training. The CoEs cover different areas, from engineering and renewable energy to Big Data science and bio-molecular research.
There are currently 14 CoEs that got selected by the European Commission under different research infrastructure calls: BioExcel, ChEESE, CoEC, CompBioMed, E-CAM, EoCoE, ESiWACE, EXCELLERAT, HiDALGO, MaX, NOMAD, PerMedCoE, POP2, and TREX
Focus CoE is contributing to the success of the European HPC ecosystem and the EuroHPC Initiative by supporting the CoEs to more effectively fulfil their role within the ecosystem and initiative: ensuring that extreme scale applications result in tangible benefits for addressing scientific, industrial or societal challenges.
Focus CoE is doing this by creating an effective platform for the CoEs to coordinate strategic directions and collaboration. It addresses possible fragmentation of activities across the CoEs and coordinates interactions with the overall HPC ecosystem. It provides support services for the CoEs in relation to both industrial outreach and promotion of their services and competences by acting as a focal point for users to discover those services.
Meet the FocusCoE consortium
The FocusCoE project consortium consists of 12 partners:

scapos AG, Germany

Commissariat a l'Energie Atomique et aux Energies Alternatives (CEA), France

Kungliga Tekniska Högskolan, Sweden

High-Performance Computing Center, University of Stuttgart, Germany

Barcelona Supercomputing Center - Centro Nacional de Supercomputacion, Spain

University College London, UK

Agenzia nazionale per le nuove tecnologie, l'energia e lo sviluppo economico sostenibile (ENEA)

National University of Ireland

Teratec, France

Forschungszentrum Jülich GmbH, Germany

Partnership for Advanced Computing in Europe AISBL

